Delivering Novel Therapies for RAS/MAPK Pathway Driven Cancers

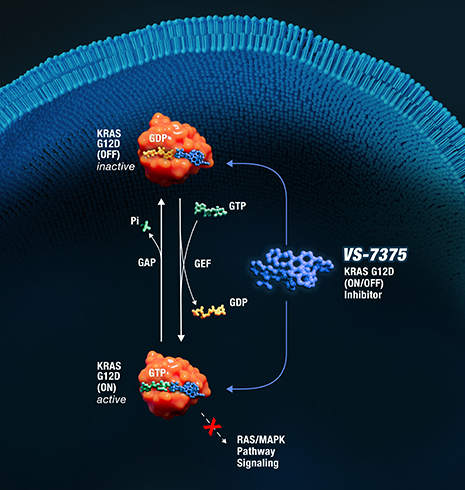
Selectively targeting the most prevalent KRAS mutation in human cancers
KRAS G12D represents 26% of all KRAS mutations, making it the most prevalent KRAS mutation in human cancers.
Advancing therapies that directly target KRAS G12D may help address a significant unmet need across multiple solid tumor types.

Precision targeting of RAS/MAPK pathway-driven cancers
Our pipeline is powered by ongoing trials and strategic collaborations centered on novel small molecule inhibitors targeting the critical RAS/MAPK signaling pathway known to drive cell survival and tumor growth. This includes targeting RAS directly with KRAS G12D inhibition, targeting the pathway downstream with RAF/MEK inhibition, and targeting the parallel pathway that drives resistance with FAK inhibition.
Get our latest news and updates
Stay informed on key announcements, the latest clinical developments, and more.



